|
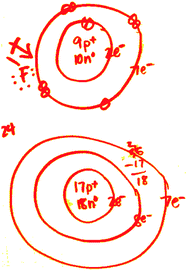
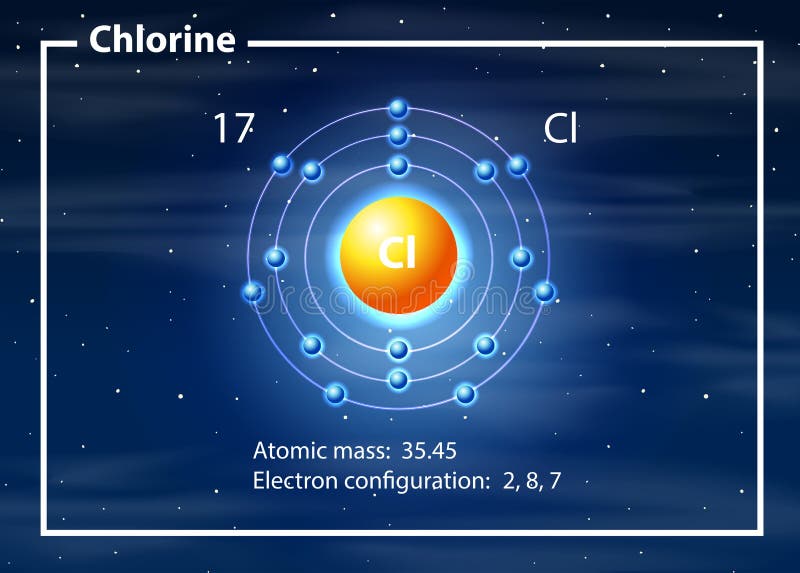
The radius increases sharply between the noble gas at the end of each period and the alkali metal at the beginning of the next period. For instance, the radii generally decrease rightward along each period (row) of the table, from the alkali metals to the noble gases and increase down each group (column). Ītomic radii vary in a predictable and explicable manner across the periodic table. Under some definitions, the value of the radius may depend on the atom's state and context. Depending on the definition, the term may apply only to isolated atoms, or also to atoms in condensed matter, covalently bound in molecules, or in ionized and excited states and its value may be obtained through experimental measurements, or computed from theoretical models. Since the boundary is not a well-defined physical entity, there are various non-equivalent definitions of atomic radius. 188 pm (Van der Waals).The atomic radius of a chemical element is the distance from the center of the nucleus to the outermost shell of an electron. Here are a few diverse ones: Calculated Atomic Radii (pm)Argon is a chemical element with symbol Ar and atomic number 18. I see many images online where argon’s atomic radius is smaller than that of chlorine. Relative atomic radius is mostly determined by. Actually, Cl has a greater atomic radius than Ar because of the greater attraction from the nucleus in Ar. Atomic Symbol: Ar, Melting Point: Atomic Weight: 39.95, Boiling Point: Electron Configuration: 3s23p In simpler terms, atomic radius of argonĪnswer: Actually, Cl has a greater atomic radius than Ar because of the greater attraction from the nucleus in Ar. Ionic Radius Atomic radius is generally stated as being the total distance from an atom’s nucleus to the outermost orbital of electron. The atomic radius is the distance from the nucleus of an atom to the. Data taken from John Emsley, The Elements, 3rd edition. In other words, it is the distance from the center of. Atomic radius is the distance from the centre of the nucleus to the outermost shell containing electrons. atomic radius, half the distance between the nuclei of identical neighbouring atoms in the solid form of an element. The atomic radius is measured as half the distance between two nuclei of the same atoms that are bonded together.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
